Moderna Booster Approval
1 a few days after Pfizer and BioNTech submitted their booster data. Fully vaccinated for COVID-19 with the Moderna vaccine.
Covid Booster Shots Everything You Need To Know The Brink Boston University
Moderna announced on Wednesday it has completed its submission to the US Food and Drug Administration for full approval of its Covid-19 vaccine.

Moderna booster approval. People in the recommended groups who got the Moderna or JJJanssen vaccine will likely need a booster shot. The CDC has already approved a third dose of Moderna for those with moderately to severely compromised immune systems Modernas proposed booster shot is different however. For the most up-to-date news and information about the coronavirus pandemic visit the WHO and CDC websites.
But what about people who received the Moderna or JJ vaccines. People should get a third shot of that vaccine at least six months after the second shot Moderna said. The pharmaceutical company Moderna is seeking approval to offer an additional booster shot of its Covid-19 vaccine.
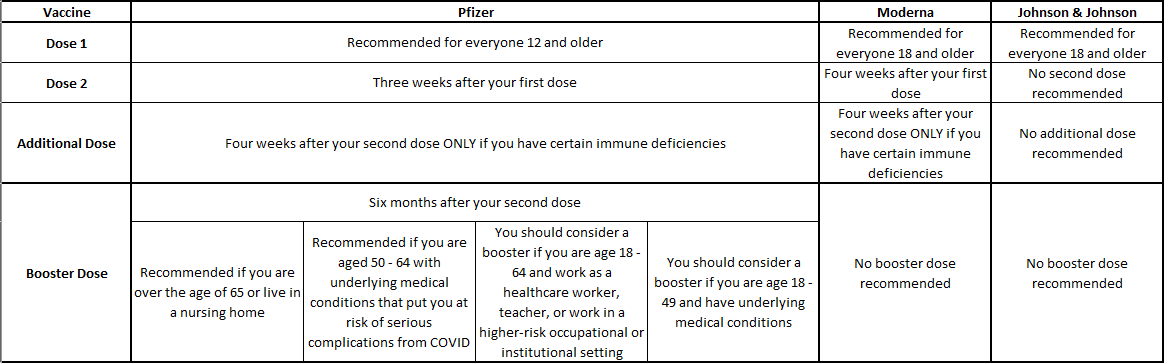
Its the Centers for Disease Control and Preventions goal that vaccinated Americans will beginning receiving a booster mRNA vaccine this fall or at least eight months after they received their second shot. If you were one of the first to get Modernas vaccine in 2020 the drug maker said on Wednesday the. At this time the Pfizer-BioNTech booster authorization only applies to people whose primary series was Pfizer-BioNTech vaccine.
12 Fauci admitted that while it was likely everyone will someday need a COVID-19 booster shot regardless of whether they originally received Pfizer or Moderna officials would only be focusing on getting extra doses to the vulnerable parts of the population for the time being. Boosters will make a bigger financial impact over the long run. FDA amended the emergency use authorization EUA for the Pfizer-BioNTech COVID-19 Vaccine to allow for the use of a single booster dose to be.
Earlier this month both the Moderna and Pfizer-BioNTech were approved as a booster for qualified people with a compromised immune system. Anthony Fauci on Tuesday said that theres a chance that Modernas COVID-19 booster shot may not have FDA and CDC approval by the White Houses Sept. If you got the Moderna vaccine against COVID-19 you could be in line for a booster shot.
Moderna submitted initial data for a booster dose on Sept. We will address with the same sense of urgency recommendations for the Moderna and JJ vaccines as. 1 day agoThe Food and Drug Administration is likely to authorize a booster dose of Modernas Covid-19 vaccine and will do so at a dose level that is half as large as what was used for the prime doses.
20 deadline for booster approval. Pfizer-BioNTech has also asked for authorization for a booster. Modernas COVID-19 booster vaccine.
The decision comes as we wait for full FDA approval of the Moderna and Johnson Johnson. Ad Do not wait to be protected. Given that there was an additional week gap between the two companies requesting approval its reasonable that it could take a little longer for the FDA to authorize Modernas booster.
The best COVID-19 vaccine is the first one that is available to you. Moderna expected to miss Sept. On Thursday the CDC gave the okay for Pfizer booster shots for those 65 and older and at high-risk.
Moderna awaits FDA decisions on full approval for its COVID-19 vaccine and for booster doses. We dont feel at. Top health officials told the White House they need more time to review Modernas application because data was inadequate and.
FDA could authorize Moderna COVID booster vaccine at a half dose. Today ACIP only reviewed data for the Pfizer-BioNTech vaccine. The company has asked the European Medicines Agency EMA in Amsterdam to approve the product for the additional vaccination.
Moderna made the announcement in a press release but the research has not yet been peer-reviewed. All authorized COVID-19 vaccines are effective. More data on the effectiveness and safety of Moderna and JJJanssen booster shots are expected soon.
Approval status who would get it and more. This aligns with the FDAs booster authorization and makes these groups eligible for a booster shot. MRNA announced that it has submitted a regulatory application with the European Medicines Agency EMA seeking conditional marketing approval for the third or booster.
During an interview on CBS This Morning on Aug. The FDA approved a third booster shot for people over age 65 and others at increased risk who have received the Pfizer vaccine.

Moderna Covid Vaccine Booster Produces Robust Response Against Delta

Moderna Submits Preliminary Data To Fda For Covid 19 Booster Shot

Timelines For F D A Approval For Moderna And J J Vaccines Remain Unclear The New York Times

Moderna Submits Data To Fda For Covid Booster Approval

Moderna Working On Hybrid Covid 19 Flu Vaccine

Moderna Requests Ema Approval For Extra Covid Vaccine Booster Shot Nl Times
Moderna Expected To Miss Sept 20 Deadline For Booster Approval
Moderna Asks Fda To Authorize A Booster Shot Of Its Covid 19 Vaccine Coronavirus Updates Npr

Moderna Seeks Fda Approval For Booster Shots Of Its Covid 19 Vaccine Healthcare Finance News

Beaumont Doctor Expects Moderna Vaccine To Get Fda Approval Followed By Booster

Moderna Ceo Says Enough Covid Vaccine Will Be Available To Return To Normal In A Year Masslive Com






Post a Comment for "Moderna Booster Approval"